What makes a promising scientific asset truly fundable is whether it is operational, whether the path from milestone to milestone is robust.
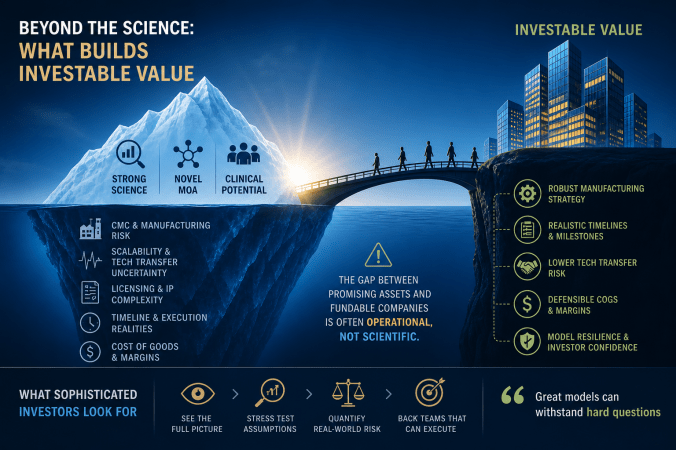
Designing Biotech Financial Models for Series B Success from WorldPharmaToday reframes what gets scrutinized in biotech financial models. Here are the key takeaways:
- CMC is now a financial variable
- Investors recognize that CMC can explain why most efficacious assets do not survive long enough to receive regulatory blessing or generate predicted revenue
- We can add clarity to seemingly nebulous predictions by examining the CMC challenges and how a company deals with them
- Go beyond clinical milestones toward the manufacturing timelines that inform the availability of clinical trial material. Merely assuming that clinical trial material will be available on demand is folly, especially when there are signs available to make more intelligent predictions.
- Do we know all the challenges that the drug product will face? Do our preformulation experimental results increase our confidence?
- Does our chosen CDMO have or can absorb the equipment necessary for manufacturing? Are the floor operators adequately trained?
- Does the CDMO have the resources to test and release raw materials and drug products? If not, do we have external testing alternatives?
- Weak assumptions quietly break models
- Over-optimistic IND timelines, vague CDMO plans, and incomplete licensing stacks can affect credibility
- Deals can be hampered by scale-up failures, tech transfer delays, and hidden royalties
The best models can hold steady upon thorough examination.